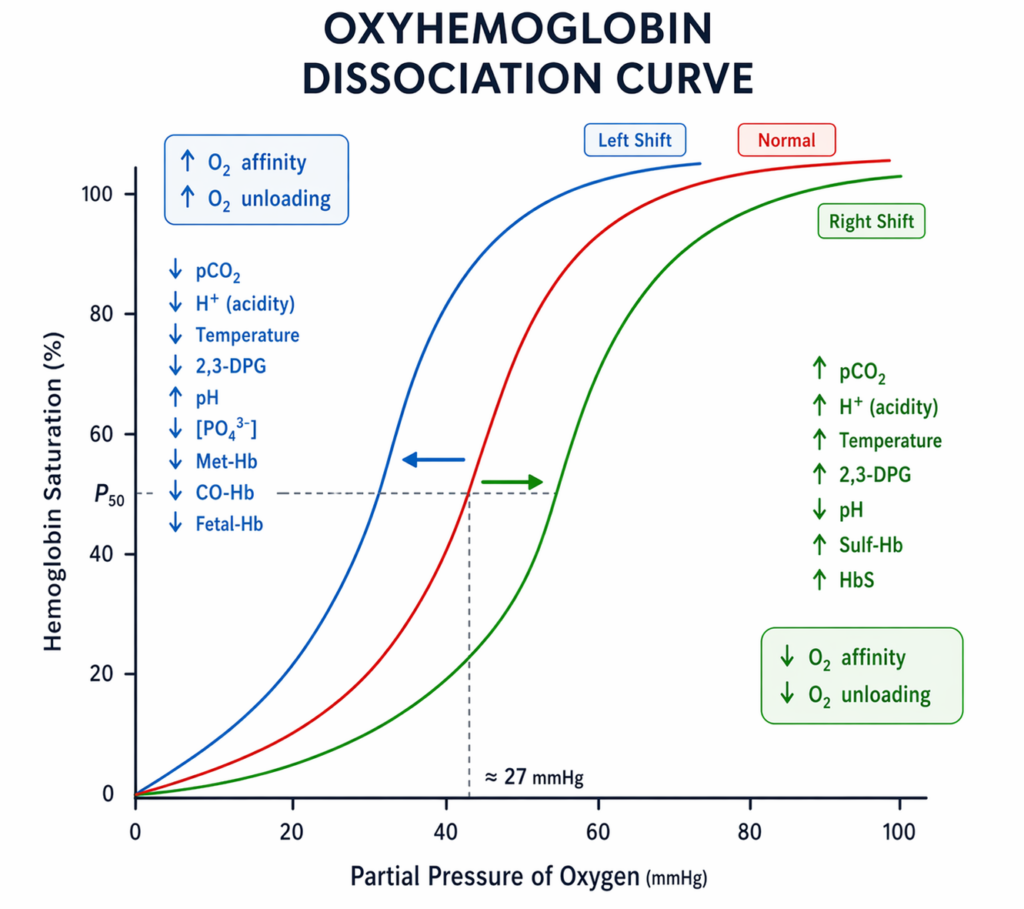
Definition and shape
- ODC describes relationship between oxygen partial pressure (PO2) and haemoglobin oxygen saturation (SO2).
- Sigmoid shape due to cooperative binding: binding of one O2 increases affinity for subsequent O2 molecules (T state → R state transition).
- Plateau around PaO2 ~8 kPa (60 mmHg): above this, saturation changes little with PaO2, below this, saturation falls rapidly.
Key numerical values (adult HbA, standard conditions)
- Standard conditions for reference curves:
- 37°C
- pH 7.40
- PaCO2 5.3 kPa (40 mmHg)
- 2,3-DPG normal
- P50:
- PO2 at which Hb is 50% saturated, HbA P50 ≈ 3.5 kPa (26–27 mmHg).
- Typical arterial values:
- PaO2 10–13 kPa (75–100 mmHg) gives SaO2 ~95–98%.
- Mixed venous:
- PvO2 ~5.3 kPa (40 mmHg) gives SvO2 ~75% (varies with VO2/DO2).
Oxygen content and delivery (linking the ODC to clinical physiology)
- Arterial oxygen content:
- CaO2 = (1.34 × Hb × SaO2) + (0.023 × PaO2) in mL O2/dL (PaO2 in kPa).
- If using mmHg: dissolved O2 term ≈ 0.003 × PaO2 (mL O2/dL).
- Dissolved O2 is usually small at normobaric pressures, Hb-bound O2 dominates.
- CaO2 = (1.34 × Hb × SaO2) + (0.023 × PaO2) in mL O2/dL (PaO2 in kPa).
- Oxygen delivery: DO2 = CO × CaO2 × 10 (if CaO2 in mL/dL, gives mL/min).
- Oxygen consumption: VO2 = CO × (CaO2 − CvO2) × 10, extraction ratio normally ~25%.
Right shift vs left shift (causes and consequences)
- Right shift = decreased Hb affinity for O2 (↑P50): promotes unloading in tissues but lowers SaO2 at a given PaO2.
- Causes: ↑H+ (↓pH), ↑PaCO2, ↑temperature, ↑2,3-DPG.
- Clinical examples: exercise, fever/sepsis, chronic hypoxaemia (via ↑2,3-DPG), anaemia (↑2,3-DPG), pregnancy (maternal).
- Left shift = increased Hb affinity for O2 (↓P50): improves loading but impairs unloading (tissue hypoxia risk).
- Causes: ↓H+ (↑pH), ↓PaCO2, ↓temperature, ↓2,3-DPG, fetal Hb (HbF), carboxyhaemoglobin, methaemoglobin.
- Clinical examples: hypothermia (peri-arrest/CPB), acute hypocapnia (over-ventilation), stored blood (low 2,3-DPG), CO poisoning.
Bohr effect and Haldane effect (often examined together)
- Bohr effect: ↑CO2/↑H+ reduces Hb affinity for O2 (right shift) enhancing O2 unloading in tissues, opposite in lungs.
- Haldane effect: oxygenation of Hb reduces its capacity to carry CO2 (as carbamino compounds) and H+, promotes CO2 unloading in lungs. Deoxygenated Hb carries more CO2.
- Together: tissues (high CO2, low O2) favour O2 unloading and CO2 loading, lungs (high O2) favour O2 loading and CO2 unloading.
2,3-DPG and stored blood
- 2,3-DPG binds deoxy-Hb (β chains) stabilising T state → right shift (↑P50) aiding unloading.
- Stored blood: 2,3-DPG falls during storage → left shift, after transfusion, 2,3-DPG regenerates over ~24–72 hours (variable).
Fetal haemoglobin and placental oxygen transfer
- HbF (α2γ2) has higher O2 affinity than HbA (left-shifted curve, lower P50) because γ chains bind 2,3-DPG less strongly.
- Placental transfer aided by: maternal right shift (↑CO2/↓pH in maternal blood near placenta) and fetal left shift, plus the double Bohr effect (CO2 transfer to mother alkalinises fetal blood).
Dyshemoglobins: COHb and MetHb
- Carboxyhaemoglobin: CO binds Hb with ~200–250× affinity vs O2 → reduces available binding sites and causes left shift of remaining sites (impaired unloading).
- Pulse oximetry may over-read (classically trends toward ~100% early, then ~90% at high COHb depending on device), requires co-oximetry for diagnosis.
- PaO2 can be normal because it reflects dissolved O2, not Hb-bound O2.
- Methaemoglobin (Fe3+): cannot bind O2, also left-shifts remaining Fe2+ sites. Pulse oximetry tends to read ~85% regardless of true saturation (device-dependent).
- Causes: local anaesthetics (prilocaine/benzocaine), nitrates/nitrites, dapsone, treat with methylene blue if significant and not G6PD deficient.
Effects of anaesthesia and critical illness
- Hypothermia: left shift (↓P50) and reduced metabolic rate, can mask tissue hypoxia by preserving SaO2 while impairing unloading.
- Hyperventilation: ↓PaCO2/↑pH → left shift, may reduce cerebral blood flow and impair tissue O2 delivery despite high SaO2.
- Sepsis/fever/acidosis: right shift, plus microcirculatory dysfunction means SvO2 may be high despite tissue hypoxia (impaired extraction).
Clinical application of ODC
- Interpret SpO2/PaO2 in context of the ODC: small PaO2 changes on the steep part cause large saturation changes, on the flat part saturation is preserved despite moderate PaO2 falls.
- Steep part (~PaO2 2–6 kPa / 15–45 mmHg): facilitates unloading in tissues, sensitive to hypoventilation, V/Q mismatch, shunt.
- Flat part (~PaO2 >8 kPa / >60 mmHg): safety margin for loading in lungs, SpO2 may remain high despite falling PaO2 until the knee is reached.
- Differentiate oxygenation (PaO2/SaO2) from oxygen delivery (DO2): a normal SpO2 does not guarantee adequate CaO2 or DO2.
- CaO2 depends mainly on Hb concentration and saturation, PaO2 contributes little unless very high (hyperbaric).
- Anaemia: PaO2 and SaO2 may be normal but CaO2 reduced, treat cause/consider transfusion based on clinical context.
- CO poisoning: PaO2 may be normal, SpO2 may be falsely reassuring, CaO2 and tissue delivery impaired.
- Use shifts to predict tissue unloading: right shift improves unloading, left shift impairs unloading (but may improve loading).
- In sepsis/fever/acidosis: right shift helps unloading but may reduce SaO2 at a given PaO2.
- In hypothermia/alkalosis: left shift increases SaO2 for a given PaO2 but may worsen tissue hypoxia.
Test yourself…
Draw and describe the oxygen dissociation curve. What are its key features and why is it sigmoid?
Structure your answer: axes → shape → key points → mechanism (cooperativity) → physiological significance.

- Axes: PO2 (x-axis) vs Hb saturation (y-axis).
- Sigmoid curve due to cooperative binding (T state to R state transition).
- Flat upper part: maintains high saturation despite moderate falls in PaO2 (safety margin for pulmonary loading).
- Steep lower part: large changes in saturation for small changes in PO2 (facilitates tissue unloading).
- Knee around PaO2 ~8 kPa (60 mmHg).
Define P50. What is its normal value and what does a change in P50 indicate?
P50 is a compact way to describe affinity changes.
- P50 = PO2 at which Hb is 50% saturated.
- Normal adult HbA P50 ≈ 3.5 kPa (26–27 mmHg) at 37°C, pH 7.40, PaCO2 5.3 kPa.
- ↑P50 = right shift (reduced affinity, enhanced unloading). ↓P50 = left shift (increased affinity, impaired unloading).
List causes of a right shift and explain the clinical advantage and disadvantage.
Give the classic quartet then link to tissues vs lungs.
- Causes: ↑H+ (acidosis), ↑PaCO2, ↑temperature, ↑2,3-DPG.
- Advantage: improved O2 unloading at tissue PO2 (helps meet increased metabolic demand).
- Disadvantage: for a given PaO2, SaO2 is lower (may worsen arterial saturation in lung disease).
List causes of a left shift and explain why it can be harmful despite increasing SaO2.
Emphasise impaired tissue unloading and dyshemoglobins.
- Causes: alkalosis, hypocapnia, hypothermia, ↓2,3-DPG, HbF, COHb, MetHb.
- Harm: increased affinity reduces unloading → tissue hypoxia at a given perfusion/PaO2.
- In COHb/MetHb: reduced functional Hb plus left shift of remaining sites compounds impaired delivery.
Explain the Bohr effect and how it relates to the oxygen dissociation curve.
Define it and state direction of shift.
- Bohr effect: increased CO2 and H+ decrease Hb affinity for O2 → right shift.
- Mechanism: H+ and CO2 stabilise deoxy (T) state, promotes O2 unloading in metabolically active tissues.
- In lungs: lower CO2/H+ shifts left, aiding loading.
Explain the Haldane effect and distinguish it from the Bohr effect.
Candidates often mix these up—separate what changes (O2 vs CO2 carriage) and where it occurs.
- Haldane effect: oxygenation of Hb reduces its ability to carry CO2 and H+ → promotes CO2 unloading in lungs.
- Deoxygenated Hb carries more CO2 (carbamino) and buffers more H+ (enhancing CO2 carriage in tissues).
- Bohr effect: CO2/H+ change Hb affinity for O2 (shift of ODC). Haldane: O2 saturation changes CO2 carriage.
A patient has Hb 8 g/dL, PaO2 12 kPa, SaO2 98%. Explain why they may still have poor oxygen delivery. Include an equation.
Show that CaO2 is mainly Hb-dependent, not PaO2-dependent.
- CaO2 = (1.34 × Hb × SaO2) + (0.023 × PaO2) mL/dL (PaO2 in kPa).
- With low Hb, the dominant Hb-bound term falls markedly even if SaO2 is high, dissolved O2 term is small at normobaric pressures.
- DO2 = CO × CaO2 × 10, if CO cannot compensate, tissue hypoxia occurs despite normal SpO2.
Why can PaO2 be normal in carbon monoxide poisoning, and what happens to the ODC?
Separate dissolved oxygen from Hb-bound oxygen and explain left shift.
- PaO2 reflects dissolved O2 in plasma, CO does not significantly reduce dissolved O2, so PaO2 may be normal.
- CO binds Hb with very high affinity, reducing available O2 binding sites (functional anaemia).
- Remaining Hb sites show increased affinity (left shift) → impaired tissue unloading.
- Pulse oximetry can be misleading, diagnosis requires co-oximetry.
How does fetal haemoglobin differ from adult haemoglobin on the ODC, and how is oxygen transferred across the placenta?
Mention HbF structure, 2,3-DPG interaction, and the double Bohr effect.
- HbF (α2γ2) is left-shifted (higher affinity, lower P50) because γ chains bind 2,3-DPG less strongly.
- Placental transfer: maternal blood becomes more acidic/hypercapnic near placenta → maternal right shift, fetal blood becomes more alkaline as CO2 diffuses to mother → fetal left shift (double Bohr effect).
What is the effect of hypothermia on the ODC and what are the clinical implications during anaesthesia?
Link shift to tissue unloading and monitoring.
- Hypothermia causes a left shift (increased affinity, ↓P50).
- May preserve SaO2 at a given PaO2 but impairs tissue unloading, risk of tissue hypoxia despite reassuring saturations.
- Also reduces metabolic rate/VO2, partially offsetting reduced unloading, overall effect depends on perfusion and severity.
Explain the role of 2,3-DPG in shifting the ODC. What happens with stored blood?
Mechanism then clinical consequence.
- 2,3-DPG stabilises deoxy-Hb (T state) → right shift (↑P50) aiding unloading.
- Stored blood has reduced 2,3-DPG → left shift, after transfusion, 2,3-DPG replenishes over ~24–72 hours (variable).
A patient is hyperventilated during neuroanaesthesia. Predict the effect on the ODC and potential consequences for oxygen delivery.
Use PaCO2/pH changes to predict shift and then discuss tissue unloading and CBF.
- Hyperventilation → ↓PaCO2 → ↑pH → left shift (↓P50).
- Left shift impairs tissue unloading, plus hypocapnia reduces cerebral blood flow, potentially worsening cerebral oxygen delivery despite high SaO2.
0 comments
Please log in to leave a comment.